PARTICIPANT RECRUITMENT AND RETENTION
Our agile approaches to participant recruitment and retention are tailored to the study protocol and local situation to ensure rapid enrollment and robust retention rates.
WE PLAN RECRUITMENT SUCCESS INTO THE PROTOCOL AND SITE SELECTION
We have found that a fit-for-purpose trial design improves participant recruitment and retention, increases equitable access to research and allows greater agility when disease-related epidemiology changes.
To achieve this, we identify the populations who would benefit most from the intervention and then select capable, research-ready sites within those areas.
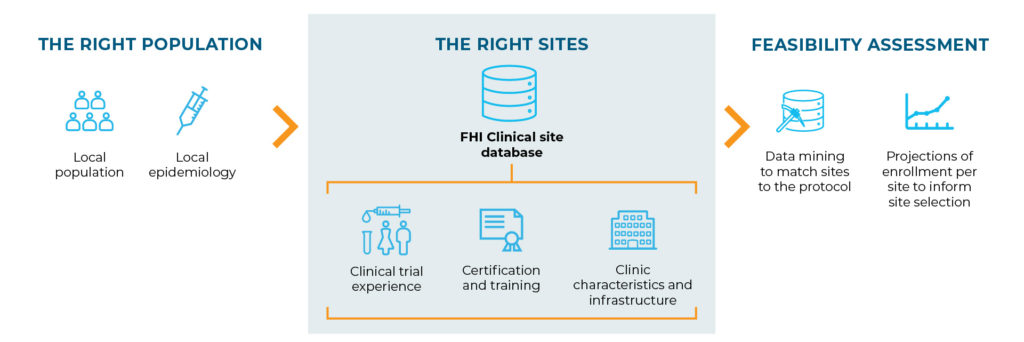
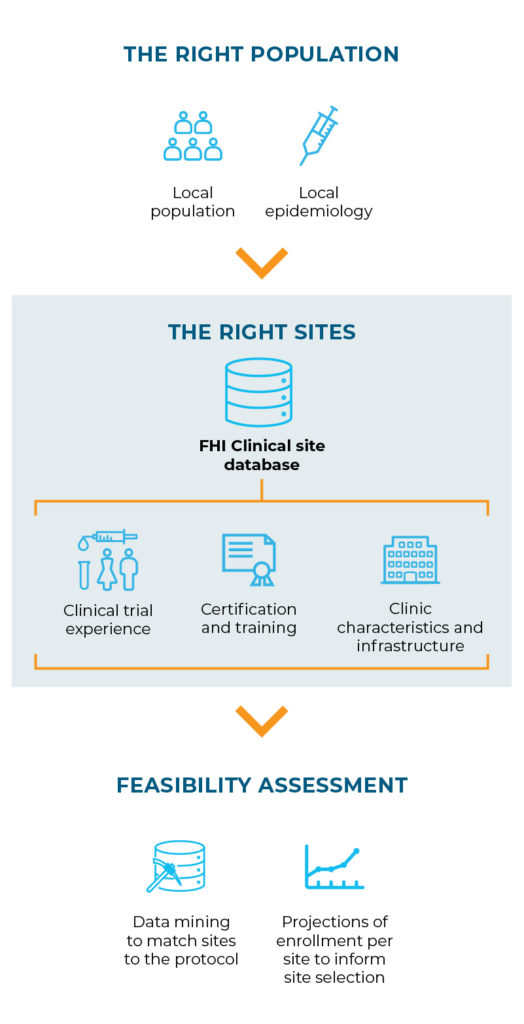
One of the benefits of this approach is being able to collect and build the ground truth into the protocol design early.
- It avoids trying to fit sites and their populations to a protocol written to achieve very specific outcomes that might not fit their situation.
- Instead, local epidemiological data are used to determine how to achieve the general study objective within the target population.
- Then, the protocol design can match the ground truth, ensuring greater success for the population, sites and study overall.
WE ADAPT TO THE LOCAL SITUATION
The study protocol, local population’s culture and disease epidemiology influence the ease and speed of participant recruitment as well as the ability to keep participants in the study. Therefore, to successfully engage participants, we approach each study with our unique set of strategies and tools based on:
Industry best practices
Local knowledge
Cultural competence
Experience across diverse populations
We build trusted, sustainable relationships with community stakeholders to:
- Engage the broader community to educate about the research purpose, benefits and risks.
- Identify and address local concerns in study design and conduct and the dissemination of research results.
- Involve the stakeholders to give the community a voice throughout the research.
COMMUNITY-FOCUSED APPROACHES
We believe that every research project should begin and end with the community.
"My role is to work with my colleagues to ensure that the principles and values of good participatory practices are present throughout every project we conduct through PREVAIL."

Barthalomew Wilson
Partnership for Research on Vaccines and Infectious Diseases in Liberia (PREVAIL) Social Mobilization and Communications (SMC) Manager, FHI Clinical, Liberia
RESPONSIVE SOLUTIONS
Our team responds quickly to recruitment and retention challenges with evidence-based, agile solutions.
- During infectious disease outbreaks, areas of new cases shift as the disease spreads. We follow the disease epidemiology to identify sites that have access to those cases and adapt recruitment targets accordingly.
- To ensure participants continued to be engaged in studies conducted within the PREVAIL network, the team implemented a team of participant trackers assigned to follow up participants. With 24 participant trackers covering Liberia, retention rates for PREVAIL studies remain high.
Watch the video to learn more about these and other recruitment and retention solutions we’ve helped sponsors implement.
WE HAVE PROVEN RESULTS
Rapidly and efficiently implement solutions for the most challenging recruitment and retention situations by leveraging our experience in some of the most complex situations around the world.
60
%
retention rate
in an oncology study challenged by poor patient enrollment and retention due to a lengthy follow-up period
81
%
retention rate
in a Chikungunya study conducted in countries in the Caribbean disrupted by political unrest and natural disasters
Learn more about our specific experience through our case studies:
Case Study
Phase II Trial of a Chikungunya Vaccine in the Caribbean
The study team maintained a 91% retention rate in a study of a Chikungunya virus (CHIKV) vaccine, despite study disruptions due to natural disasters, including Hurricane Maria, and political unrest near the sites in five Caribbean countries in a CHIKV endemic region.
Case Study
Rapid Site Assessment and Recommendations for Clinical Trials of a New Malaria Treatment
Targeted geographic areas for the development of an antimalarial drug by Novartis included malaria-endemic regions in Africa and Asia, and Novartis requested assistance identifying and assessing potential clinical sites for their planned clinical trial. Our team conducted 38 rapid, in-depth, 2.5-day site feasibility assessments in the 18 countries.
Case Study
Rescue of an Ongoing Global, Multi-Site Oncology Trial
In a Phase 3 study with a lengthy intervention for colorectal cancer, the project team reached a 70% retention rate, with steadily increasing enrollment rates, by implementing strategies to address poor patient enrollment and retention.




