Our capabilities and experience
Read more about how we help manage complex clinical research across many therapeutic areas worldwide.
Page 5 of 6
Successful Outbreak Responses Depend On Reliable Data
In this webinar, industry veterans Michelle Berrey, MD, MPH, and Claudia Christian discuss the importance of quality data for a successful outbreak response plan.
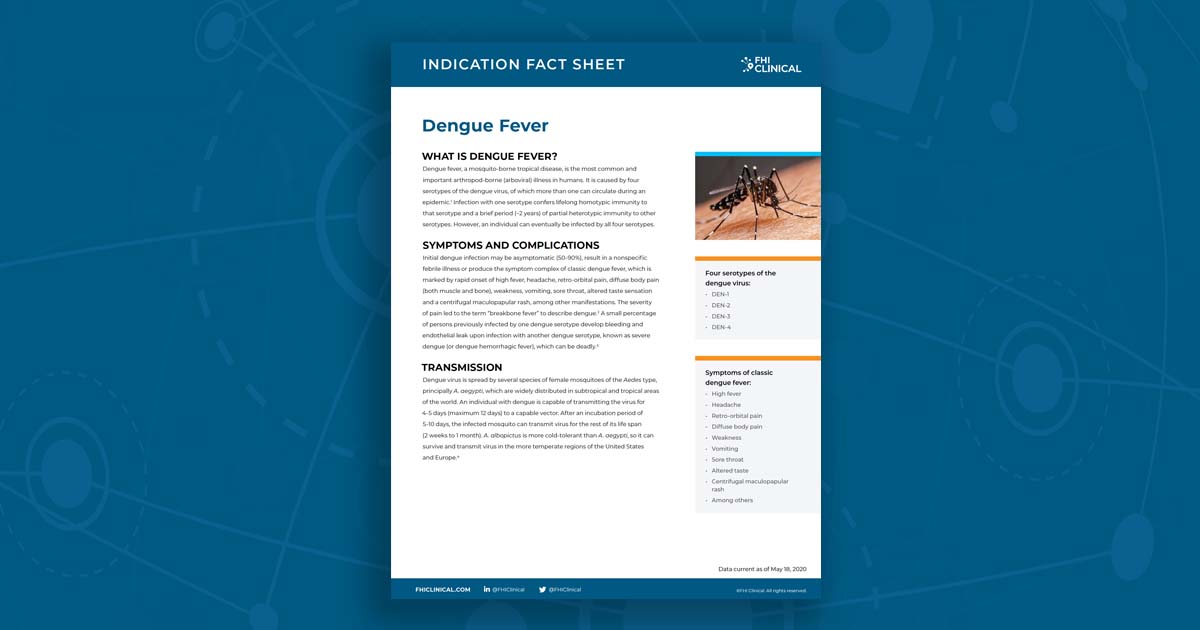
Dengue Fever Fact Sheet
Dengue fever, a mosquito-borne tropical disease, is the most common and important arthropod-borne (arboviral) illness in humans. It is caused by four serotypes of the dengue virus, of which more than one can circulate during an epidemic. Infection with one serotype confers lifelong homotypic immunity to that serotype and a brief period (~2 years) of partial heterotypic immunity to other serotypes. However, an individual can eventually be infected by all four serotypes.
8 Key Lessons for Outbreak Responses
Outbreaks large and small require rapid, coordinated responses. In this infographic, we describe eight considerations for outbreak responses, based on the collective experience of our experts.
Rapid Site Assessment and Recommendations for Clinical Trials of a New Malaria Treatment
Targeted geographic areas for the development of an antimalarial drug by Novartis included malaria-endemic regions in Africa and Asia, and Novartis requested assistance identifying and assessing potential clinical sites for their planned clinical trial. Our team conducted 38 rapid, in-depth, 2.5-day site feasibility assessments in the 18 countries.
What Can We Learn From Past Outbreaks?
Outbreaks large and small require rapid, coordinated responses. In this e-book, we describe eight considerations for outbreak responses, based on the collective experience of our experts.
Schistosomiasis Fact Sheet
Also known as snail fever, schistosomiasis is caused by parasitic flatworms called schistosomes. It is spread through contact with contaminated fresh water. Most human cases are caused by specific species of flatworms. In tropical countries, it is second only to malaria among parasitic diseases with the greatest economic impact. Schistosomiasis affects almost 200 million individuals per year, with most cases occurring in Africa, Asia and South America.
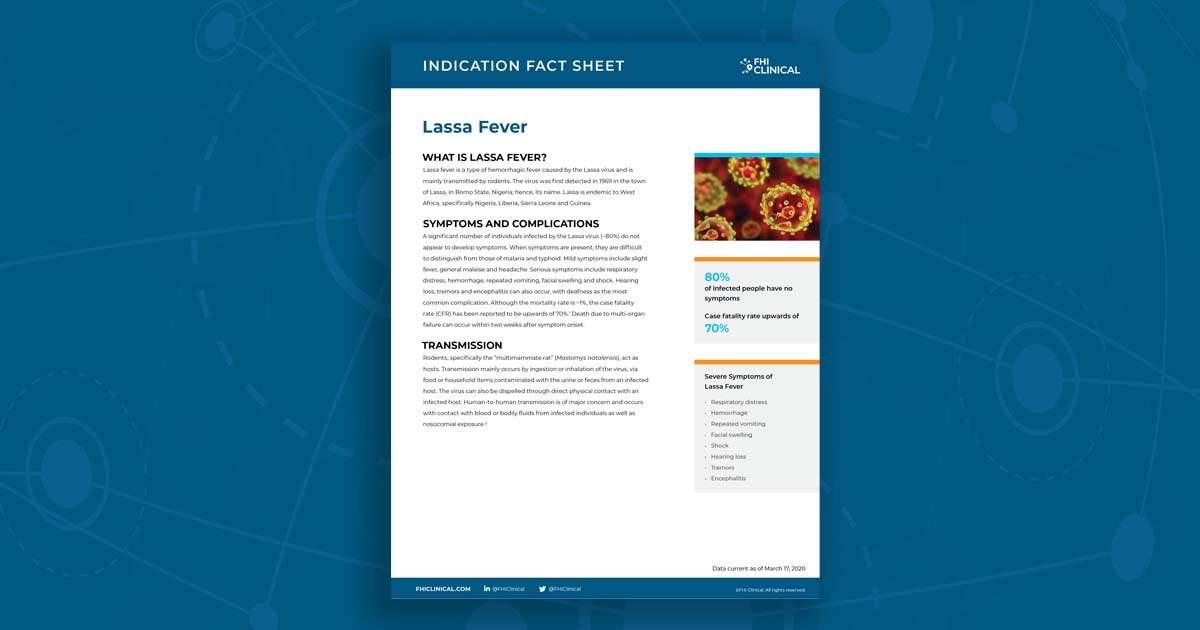
Lassa Fever Fact Sheet
Lassa fever is a type of hemorrhagic fever caused by the Lassa virus and is mainly transmitted by rodents. The virus was first detected in 1969 in the town of Lassa, in Borno State, Nigeria; hence, its name. Lassa is endemic to West Africa, specifically Nigeria, Liberia, Sierra Leone and Guinea. A significant number of individuals infected by the Lassa virus (~80%) do not appear to develop symptoms. The case fatality rate (CFR) has been reported to be upwards of 70%
Providing Site Monitoring and Management Expertise in Malaria Vaccine Trials in Equatorial Guinea
Data monitoring strategies implemented by our bilingual CRA helped eliminate the backlog to ensure timely database lock in a phase 2 trial for Sanaria’s innovative PfSPZ Vaccine for malaria in Equatorial Guinea, an area with limited prior research experience.
Research Capacity Brochure
Collaborative relationships ensure site sustainability. Keeping a site functional, even in slow times, preserves investment in human and equipment resources and ensures the site is ready to conduct quality research as soon as there is a need.
Cryptosporidiosis Fact Sheet
Cryptosporidium infection (cryptosporidiosis) is a diarrheal disease caused by tiny, one-celled Cryptosporidium parasites representing at least 15 genotypically and phenotypically diverse species. When these parasites enter the body, they travel to the distal small intestine and burrow into the intestinal walls. After 1-2 weeks, they are shed and excreted in feces. Infection can also occur in the respiratory tract.